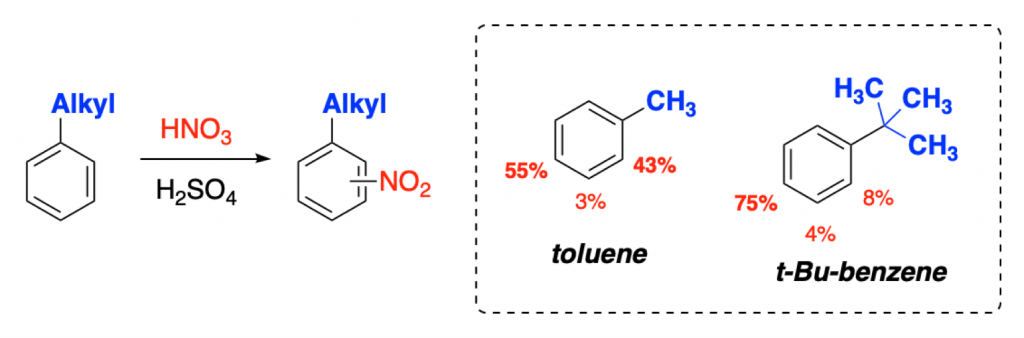
Conformational Effects on Physical-Organic Descriptors: The Case of Sterimol Steric Parameters | ACS Catalysis

Steric and Stereoelectronic Effects in Organic Chemistry: Yadav, Veejendra K.: 9789811011382: Amazon.com: Books
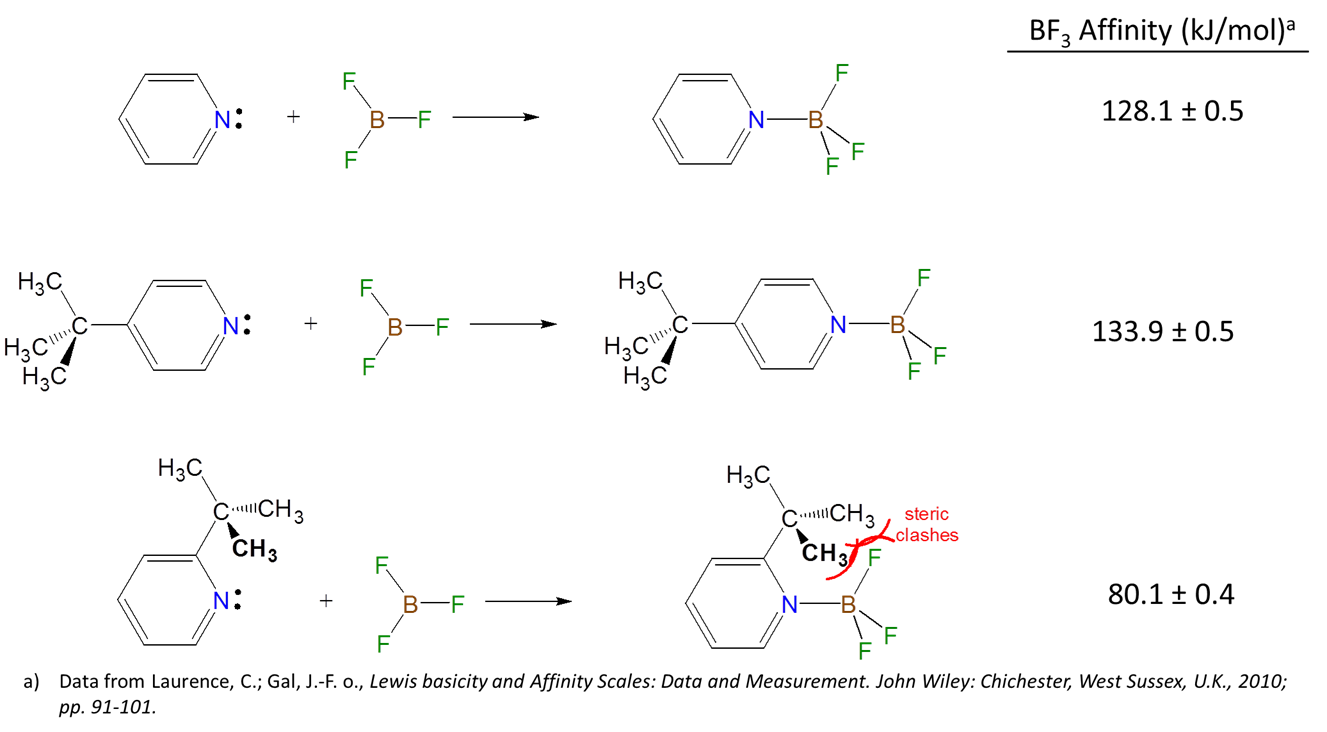
6.4.7: Bulky groups weaken the strength of Lewis acids and bases because they introduce steric strain into the resulting acid-base adduct. - Chemistry LibreTexts


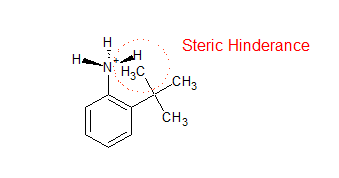

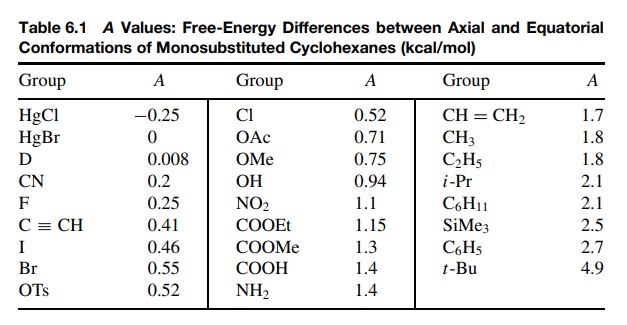




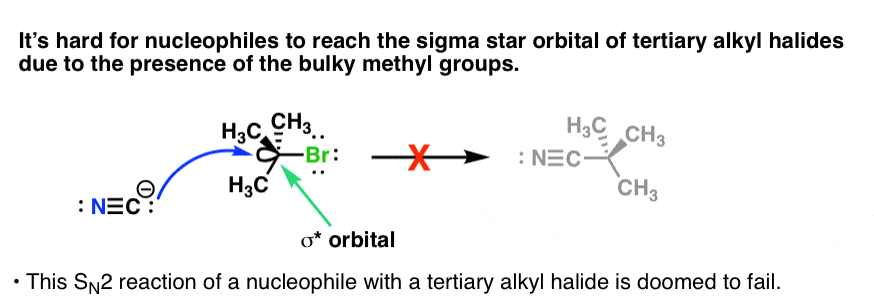


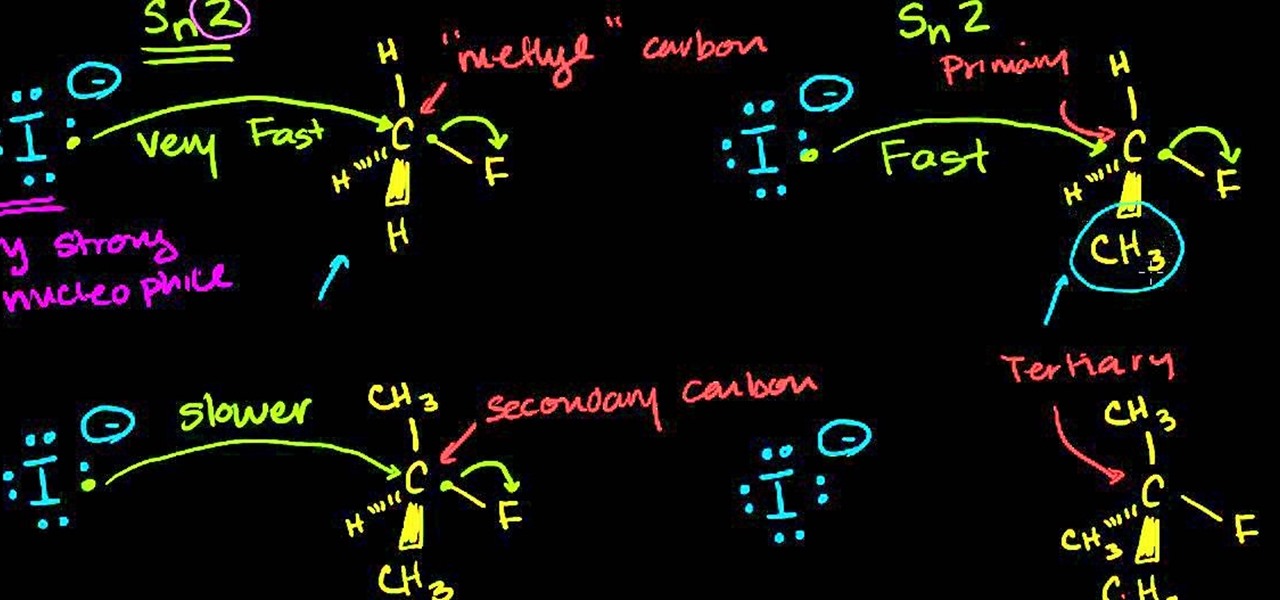

/chapter5/pages11and12/page11and12_files/substcyclohexane.png)