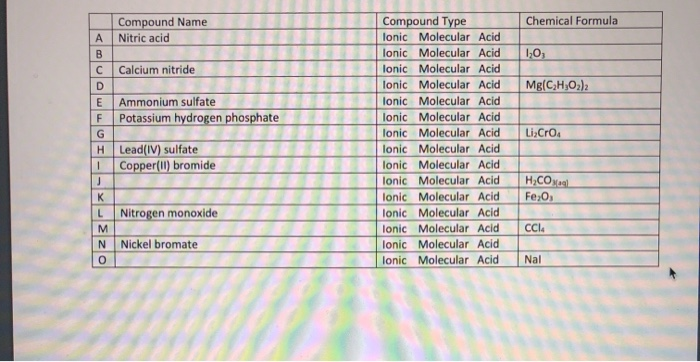
Chromate, Ion chromatography standard solution, Specpure CrO4|-2 1000 g/mL, Thermo Scientific Chemicals | Fisher Scientific

SOLVED: Balance each of the following . equations. Name any ionic compounds the reactions K;CrO4(aq) AgNOs(aq) AgCrO (s) KNOj(aq) CH;OH() O-(g) COg) H,O() FeCl; (aq) AgNOz (aq) Fe(NOw)(aq) AgCl Al(s) S(s) 1(g)

Answers (2) C (12) D (3 ) C (13) E (4) C (14) B (5) C (15) B (6) B (16) B (7) E (17) B (8) D (18) E (9) C (19) D (10) A (20) C (11) A (21) E. - ppt download

SOLVED: inhln Name each of the following polyatomic ions: CN CrO4 SO4 NO3 OH PO4 CzH3O2 NHA FORMULAS GIVEN INDIVIDUAL IONS WRITING CHEMICAL total +ve charge total ve charge Inde must be
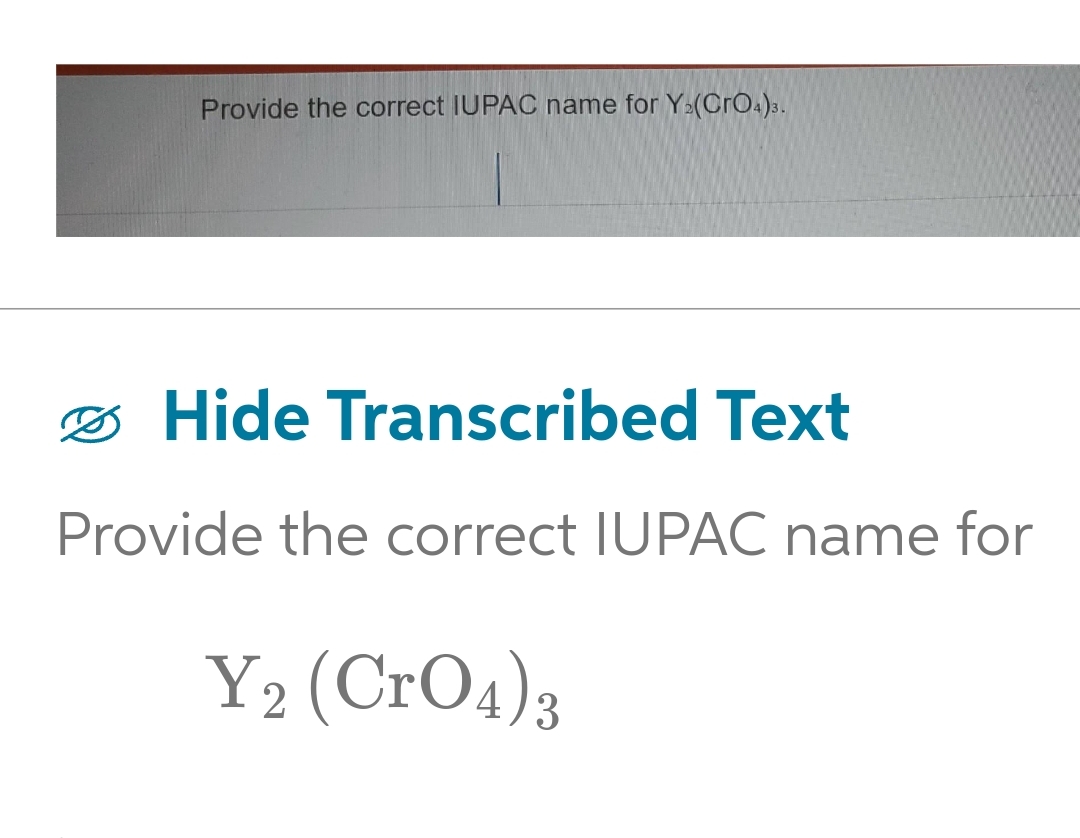
![Write the IUPAC name of the following : (a) [Cr(acac)(3)] (b) [V(H(2)O)(6)]Cl(3) ( c) (NH(4))(3)[Co(C(2)O(4))(3)] (d) K(2)[CrO(4)] Write the IUPAC name of the following : (a) [Cr(acac)(3)] (b) [V(H(2)O)(6)]Cl(3) ( c) (NH(4))(3)[Co(C(2)O(4))(3)] (d) K(2)[CrO(4)]](https://d10lpgp6xz60nq.cloudfront.net/web-thumb/644549265_web.png)
Write the IUPAC name of the following : (a) [Cr(acac)(3)] (b) [V(H(2)O)(6)]Cl(3) ( c) (NH(4))(3)[Co(C(2)O(4))(3)] (d) K(2)[CrO(4)]


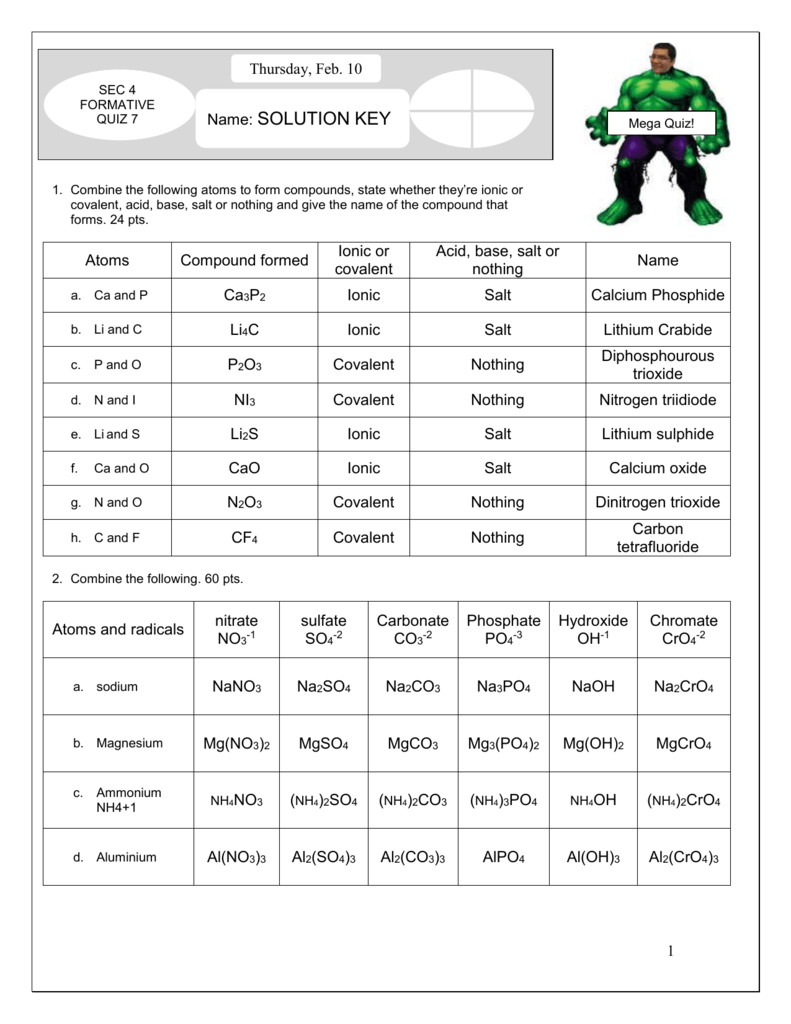


.PNG)


![ANSWERED] Name each of the following polyatomic ion... - Physical Chemistry ANSWERED] Name each of the following polyatomic ion... - Physical Chemistry](https://media.kunduz.com/media/sug-question/raw/55240199-1659276732.1714506.jpeg)


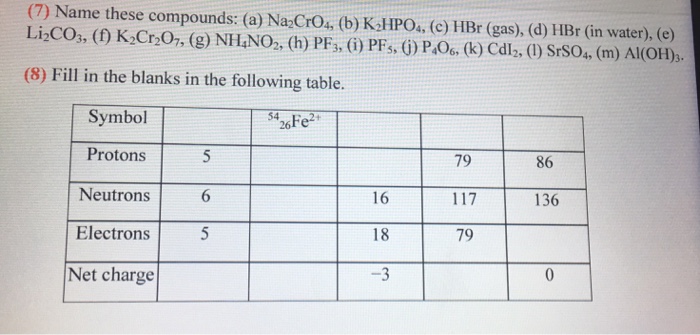
![ANSWERED] Use the References to access important va... - Physical Chemistry ANSWERED] Use the References to access important va... - Physical Chemistry](https://media.kunduz.com/media/sug-question/raw/50413524-1659108740.637296.jpeg)