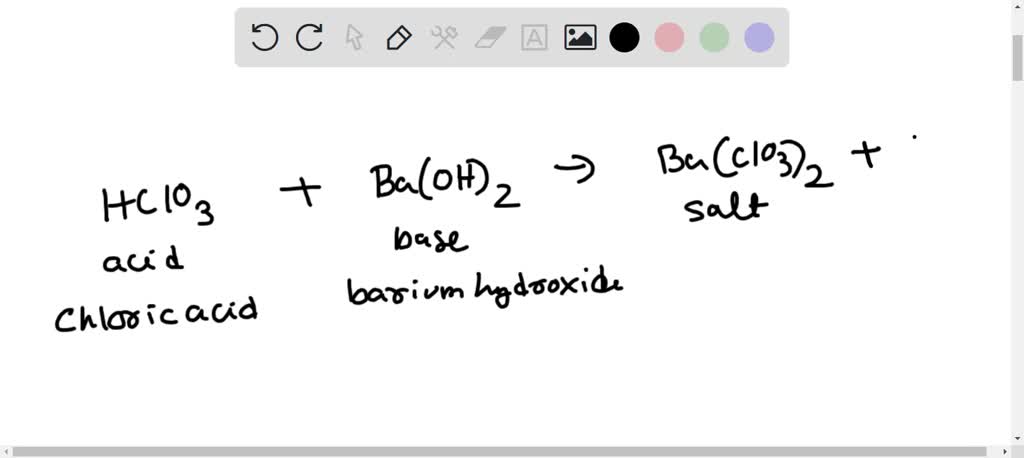
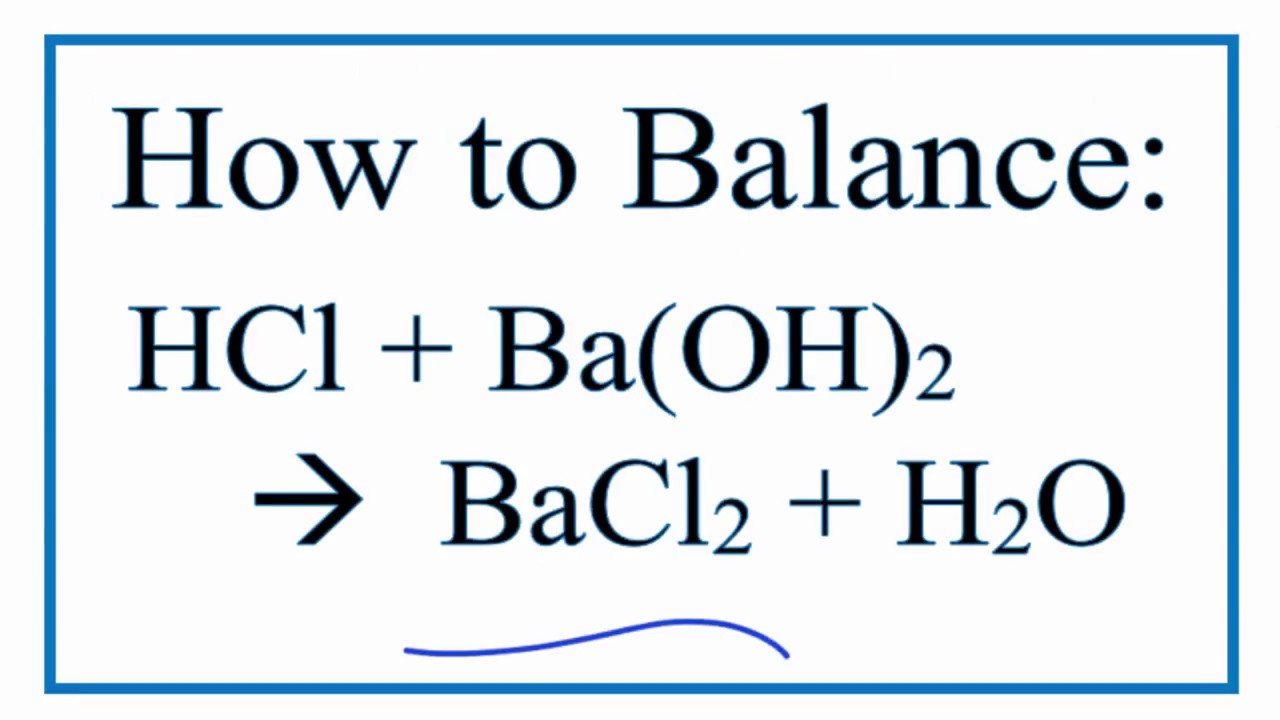
How to Write the Net Ionic Equation for Ba(OH)2 + H2SO4 = BaSO4 + H2O (Note: it should be 2H2O) - YouTube

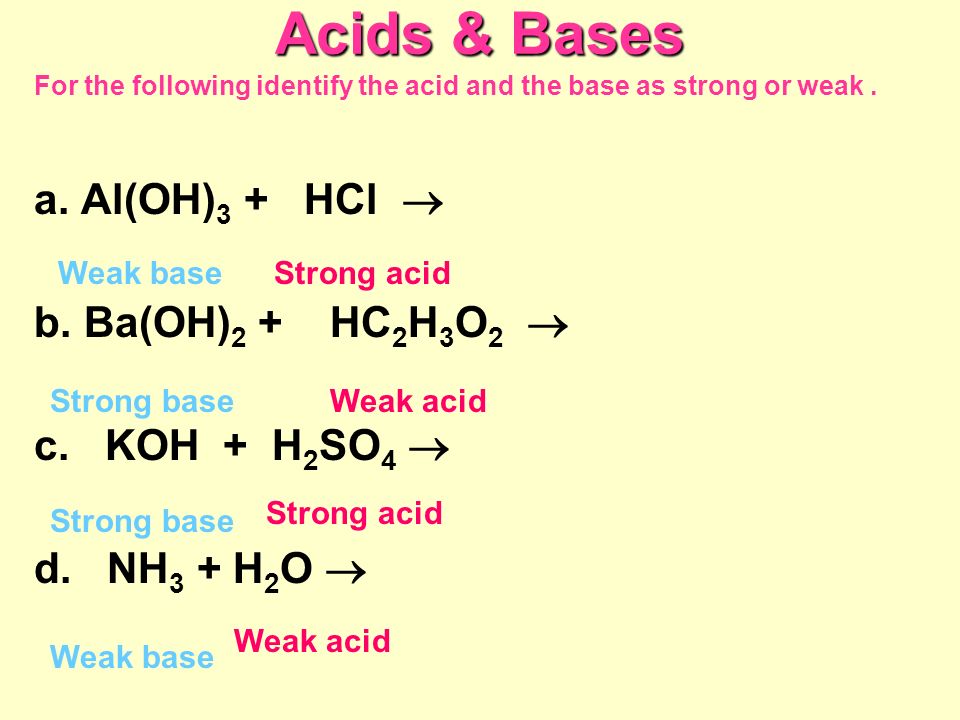
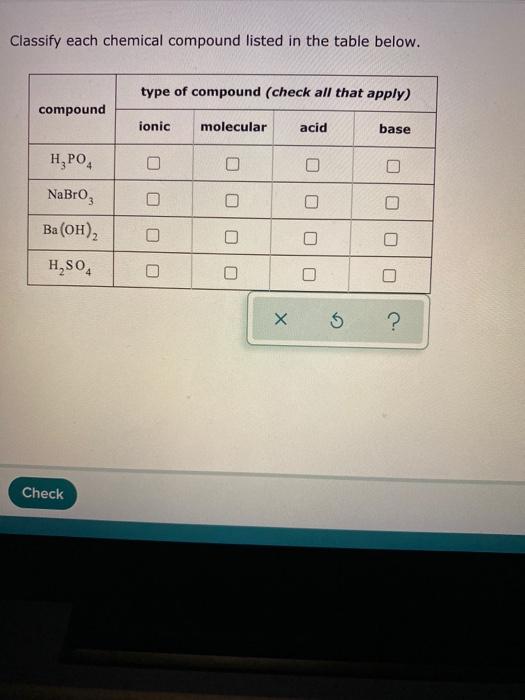
Acids & Bases Acids: acids are sour tasting Arrhenius acid Arrhenius acid: Any substance that, when dissolved in water, increases the concentration. - ppt download

Acid-Base Concepts -- Chapter Arrhenius Acid-Base Concept (last semester) Acid: H + supplier Base: OH – supplier 2. Brønsted-Lowry Acid-Base Concept. - ppt download

Question Video: Determining the Products of the Neutralization Reaction of Barium Hydroxide Ba(OH)₂ with Carbonic Acid H₂CO₃ | Nagwa