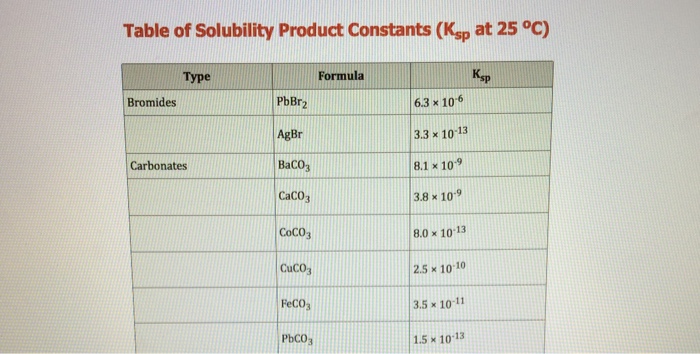
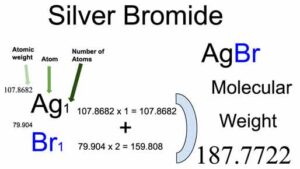
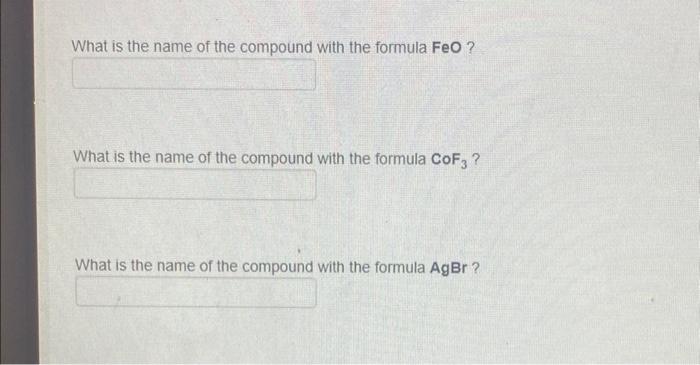
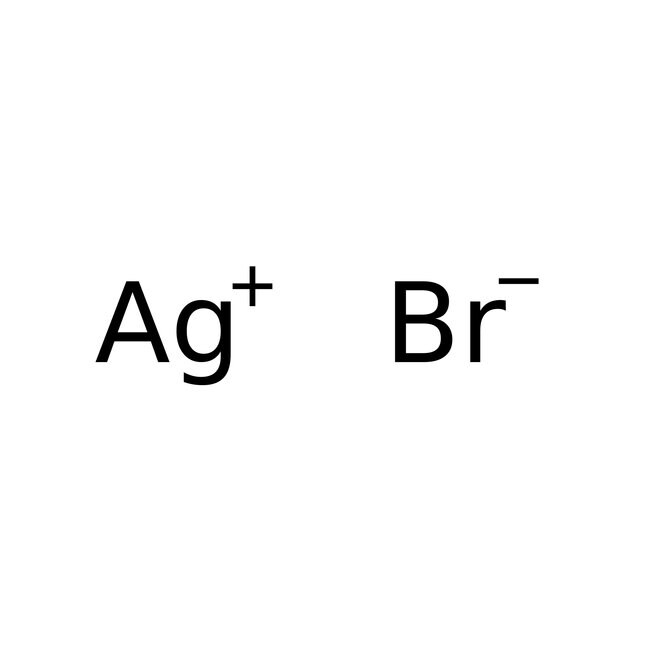SOLVED: Exercise 3. Read Naming Binary Ionic Compounds with Fixed Charge Metals (BIF) for the procedure, then name the following compounds: BaS NaH MgCl2 CaO AlCl3 Li2S ZnF2 AgBr

Silver Bromide Stock Illustrations – 6 Silver Bromide Stock Illustrations, Vectors & Clipart - Dreamstime

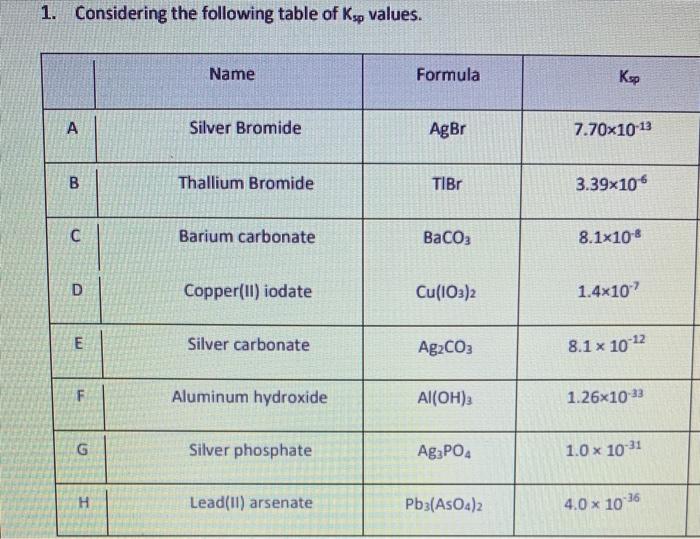
SOLVED: Which of the following compounds has the (according to the IUPAC) correct chemical name? 1. FeNO2 - Iron(II) nitrite 2. Cr(NO3)2 - Chromium(II) nitrate 3. AgBr - Silver bromide 4. ZnCl2 -